Mining and Ore Processing
Mercury Pollution
Estimated Population at Risk:
1.5 Million
Description
Mining and ore processing occurs throughout the world and consists of extracting minerals, metals, and gems from surrounding earth and ore. Industrial mines vary in size, with some being quite small operations and others very large. Many large-scale mines also have ore processing facilities where extracted ore is sent for crushing, washing, and various physical or chemical separation processes. Not all mined materials are dangerous, but many heavy metals and naturally radioactive materials that are removed from the earth can be very hazardous to human health.
In addition to active mines, there are a large number of inactive and abandoned mining sites throughout the world that still contain large levels of contaminants and pose dangers to local communities.
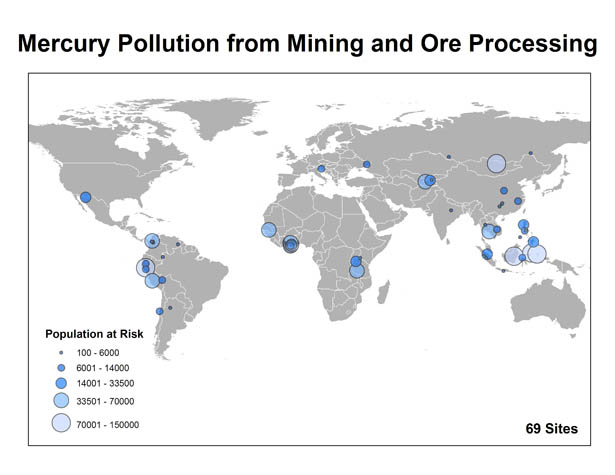
Populations estimates are preliminary and based on an ongoing global assessment of known polluted sites.
Industrial Process
 Elemental mercury occurs naturally in the earth and is a liquid metal. Most mercury forms in a sulfide ore called cinnabar, but mercury is also frequently found in small amounts in other ores. A common method for separating mercury from cinnabar is to crush the ore and then heat it in a furnace in order to vaporize the mercury. This vapor is then condensed into liquid mercury form. If done improperly, mercury vapor, which is highly toxic, can escape into the atmosphere.
Elemental mercury occurs naturally in the earth and is a liquid metal. Most mercury forms in a sulfide ore called cinnabar, but mercury is also frequently found in small amounts in other ores. A common method for separating mercury from cinnabar is to crush the ore and then heat it in a furnace in order to vaporize the mercury. This vapor is then condensed into liquid mercury form. If done improperly, mercury vapor, which is highly toxic, can escape into the atmosphere.
Waste rock and tailings from mercury mining, and other extraction processes where mercury is uncovered, can still contain small or large amounts of the toxic substance. Mercury that is not processed or claimed during mining and ore processing can make its way into the environment if the mining waste is not stored properly.
Mercury is also extensively used in the gold mining process as an amalgam for separating gold from ore. When mercury is combined with gold silt, it causes a reaction that separates the gold from the ore. This amalgam is then heated in order to evaporate the mercury and leave the gold pieces. If done without the proper safety equipment, this process can release mercury vapor into the air and can also discharge various amounts of liquid mercury into the surrounding area.
Global Context
 Mercury is uncovered and used in a number of mining processes. It is mined as a primary element for use in materials such as thermometers and fluorescent light bulbs, it can be commonly found in the ore of other mined materials, and it is frequently used in the gold mining process as an amalgam for the separation of gold from ore. Thus, mercury contamination is associated with a large variety of different mining and ore processing activities throughout the world. Due to its large health risk, international negotiations are ongoing to globally ban the use of mercury in the years to come (Mercury Convention).
Mercury is uncovered and used in a number of mining processes. It is mined as a primary element for use in materials such as thermometers and fluorescent light bulbs, it can be commonly found in the ore of other mined materials, and it is frequently used in the gold mining process as an amalgam for the separation of gold from ore. Thus, mercury contamination is associated with a large variety of different mining and ore processing activities throughout the world. Due to its large health risk, international negotiations are ongoing to globally ban the use of mercury in the years to come (Mercury Convention).
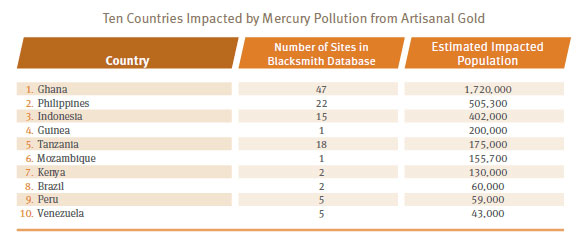
Currently, the Blacksmith inventory estimates that over 1.5 million people are affected by mercury pollution from mining and ore processing, with most pollution problems of this nature occurring in Southeast Asia and Africa.
Exposure Pathways
 Waste rock and tailings that are created during the mining and ore refining process can release toxins into the environment if not stored or disposed of properly. In many cases, waste rock and tailings are left out in the open where they are exposed to the elements, and mercury can be washed into water systems by rainfall, or can leach into the soil. Mercury that is used as an amalgam in gold mining can also be released into the environment as vapor and elemental mercury. When mercury enters water systems, it can bioaccumulate in the bodies of aquatic animals, where it becomes organic, or methylmercury. When ingested, methylmercury can be very hazardous to human health.
Waste rock and tailings that are created during the mining and ore refining process can release toxins into the environment if not stored or disposed of properly. In many cases, waste rock and tailings are left out in the open where they are exposed to the elements, and mercury can be washed into water systems by rainfall, or can leach into the soil. Mercury that is used as an amalgam in gold mining can also be released into the environment as vapor and elemental mercury. When mercury enters water systems, it can bioaccumulate in the bodies of aquatic animals, where it becomes organic, or methylmercury. When ingested, methylmercury can be very hazardous to human health.
Health Effects
The health hazards that result from exposure to mercury depend on the level of exposure and the way in which the pollutant enters the body. Inhalation of mercury vapor is particularly hazardous for kidneys, the central nervous system, and the respiratory and cardiovascular systems.15 Inhalation of mercury vapor has also been found to cause neurobehavioral disorders, such as hand tremor and mental retardation. Exposure to other forms of mercury – and in particular the methylmercury that accumulates in fish – can also lead to problems with the kidneys, lungs, and central nervous system, in addition to arthritis, reproductive problems, loss of memory, psychosis, and in some cases, death. Children exposed to mercury contamination have a higher risk of developmental complications.
What is Being Done
 An important step in curbing the pollution created by mining and ore processing is to work with mines and governments to improve mining equipment and environmental regulation. Many pollution problems occur due to a lack of set environmental and health standards, or, when laws do exist, weak enforcement and compliance procedures. However, if local governments and organizations put pressure on mines to decrease their emissions, this can be effective in terms of getting companies to institute new policies or set up new infrastructure, such as wastewater treatment facilities or more effective tailing storage areas.
An important step in curbing the pollution created by mining and ore processing is to work with mines and governments to improve mining equipment and environmental regulation. Many pollution problems occur due to a lack of set environmental and health standards, or, when laws do exist, weak enforcement and compliance procedures. However, if local governments and organizations put pressure on mines to decrease their emissions, this can be effective in terms of getting companies to institute new policies or set up new infrastructure, such as wastewater treatment facilities or more effective tailing storage areas.
Example – DALY Calculations
A small town in the mountains of Chile has been the site of large-scale gold and copper mining since 1891. Air pollution in this region is so severe that the government classified the town as a “Saturated Zone” – indicating the dangers of breathing the air around the mine. Samples taken around the site found 100 micrograms per cubic meter of mercury in the air, which is 100 times higher than the WHO health standard. Blacksmith estimates that 20,000 people may be affected by this pollution.
DALYs associated with adverse health impacts from atmospheric mercury exposure at this site are estimated to be 716,950 for the estimated exposed population of 20,000. This means that the approximately 20,000 affected people will have a collective 716,950 years lost to death, or impacted by disease or disability. This comes out to 28.7 years lost or lived with a disability per person.
Footnotes:
[15]: “Toxicological Profile for Mercury.” U.S. Department of Health and Human Services. March, 1999. Available at: http://www.atsdr.cdc.gov/toxprofiles/tp.asp?id=115&tid=24.

-
Artisanal Gold Mining
Mercury Pollution
-
Industrial Estates
Lead Pollution
-
Agricultural Production
Pesticide Pollution
-
Lead Smelting
Lead Pollution
-
Tannery Operations
Chromium Pollution
-
Mining and Ore Processing
Mercury Pollution
-
Mining and Ore Processing
Lead Pollution
-
Lead-Acid Battery Recycling
Lead Pollution
-
Arsenic in Groundwater
Arsenic Pollution
-
Pesticide Manufacturing and Storage
Pesticide Pollution
-
The Rest of the Toxic Twenty



